 I would like to confirm a flight to Medan.Ĭ. Can I book a table for tomorrow afternoon?ī. Receptionist : When would you like to go sir?Ī. Receptionist: Good afternoon Wijaya Air, can I help you? takes care of the cleanliness of the kitchenĤ. carrying away the dirty dishes to the pantryī. Lala : Dian, you are a chef in an Italian restaurant, aren’t you? What are your duties exactly?ĭian : I supervise the preparation and the service of the food from the kitchen to the dining room, arrange the menu, and.Ī. If he hadn’t broken his leg, I’m sure he would have. David : Did Ken join the futsal competition last week?Ĭlare : No, he broke his leg, so he didn’t take part of the contest. Receptionist : Family restaurant, can I help you?īraga : Yes Please. Soal latihan bahasa Inggris ini dapat kalian pergunakan juga ketika akan menghadapi ujian sekolah sebagai bahan ajar sekaligus pembelajaran tambahan.ĭibawah ini adalah Soal UAS/PAS 1 Mata Pelajaran Bahasa Inggris Kelas 12 (SMA/SMK) dengan jumlah 50 soal pilihan yang harus kalian beri tanda silang (x) pada abjad a,b,c,d dan e dengan jawaban yang menurut kalian benar dan tepat !ġ. Reactions between acids and bases that are both weak may result in solutions that are neutral, acidic, or basic.Sumber materi soal UAS/PAS 1 B Inggris ini berasal dari file soal-soal ulangan harian, uas, ukk dan berdasarkan kurikulum 2013. The products of the reaction do not have the characteristics of either an acid or a base. 
When equal amounts of a strong acid such as hydrochloric acid are mixed with a strong base such as sodium hydroxide, the result is a neutral solution. A salt is essentially any ionic compound that is neither an acid nor a base. A salt is an ionic compound composed of a cation from a base and an anion from an acid. The aqueous sodium chloride that is produced in the reaction is called a salt. 
Soal un fisika medan magnet dan pembahasan plus#So with a strong acid and strong base, the net ion equation is always H+ plus OH- yields H2O.Ī neutralization reaction is a reaction in which an acid and a base react in an aqueous solution to produce a salt and water. So the Na+, and the Cl – are spectator ions. So what you end up with is you always end up H+ plus OH- yields H2O, because HCl, and NaOH, since they’re strong, they fully dissociate. What is the net ionic equation for the reaction of any strong acid with a soluble strong hydroxide base such as NaOH? Which is the neutralization reaction?Ī neutralization reaction is a reaction in which an acid and a base react in an aqueous solution to produce a salt and water. The overall equation for this reaction is: NaOH + HCl → H2O and NaCl. Water is always one product, and a salt is also produced. That’s because the reaction produces neutral products. Reactions of Acids and Bases When an acid and a base react, the reaction is called a neutralization reaction. Soal un fisika medan magnet dan pembahasan how to#You might be interested: FAQ: How to cook fresh garbanzo beans? What is a neutralization reaction equation? 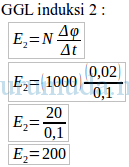
The net ionic equation is a chemical equation which gives the ions that are participated in the formation of the final product. What is ionic and net ionic equations?Ĭomplete ionic equation is a chemical equation that explains the chemical reaction, clearly indicating the ionic species present in a solution. If the acid and base are equimolar, the pH of the resulting solution can be determined by considering the equilibrium reaction of HB⁺ with water. When a weak base and a strong acid are mixed, they react according to the following net-ionic equation: B(aq) + H₃O⁺(aq) → HB⁺(aq) + H₂O(l). Which is a net ionic equation for the neutralization reaction of a strong acid with a weak base? The neutralization of a strong acid and strong base has a pH equal to 7. What is the reaction of acid base neutralization?Ī neutralization reaction is when an acid and a base react to form water and a salt and involves the combination of H+ ions and OH– ions to generate water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
